R&D Center in China
Key R&D Capabilities
Long-acting and Extended Release Technology
Liposome and Targeted Drug Delivery Technology
New Molecular Entity Platforms
Biological Antibody Platforms
R&D Center in Europe
Key R&D Capabilities
Transdermal Drug Delivery Technology
Granted Patents
251 patents granted in China
577 patents granted overseas
(As of December 2025)

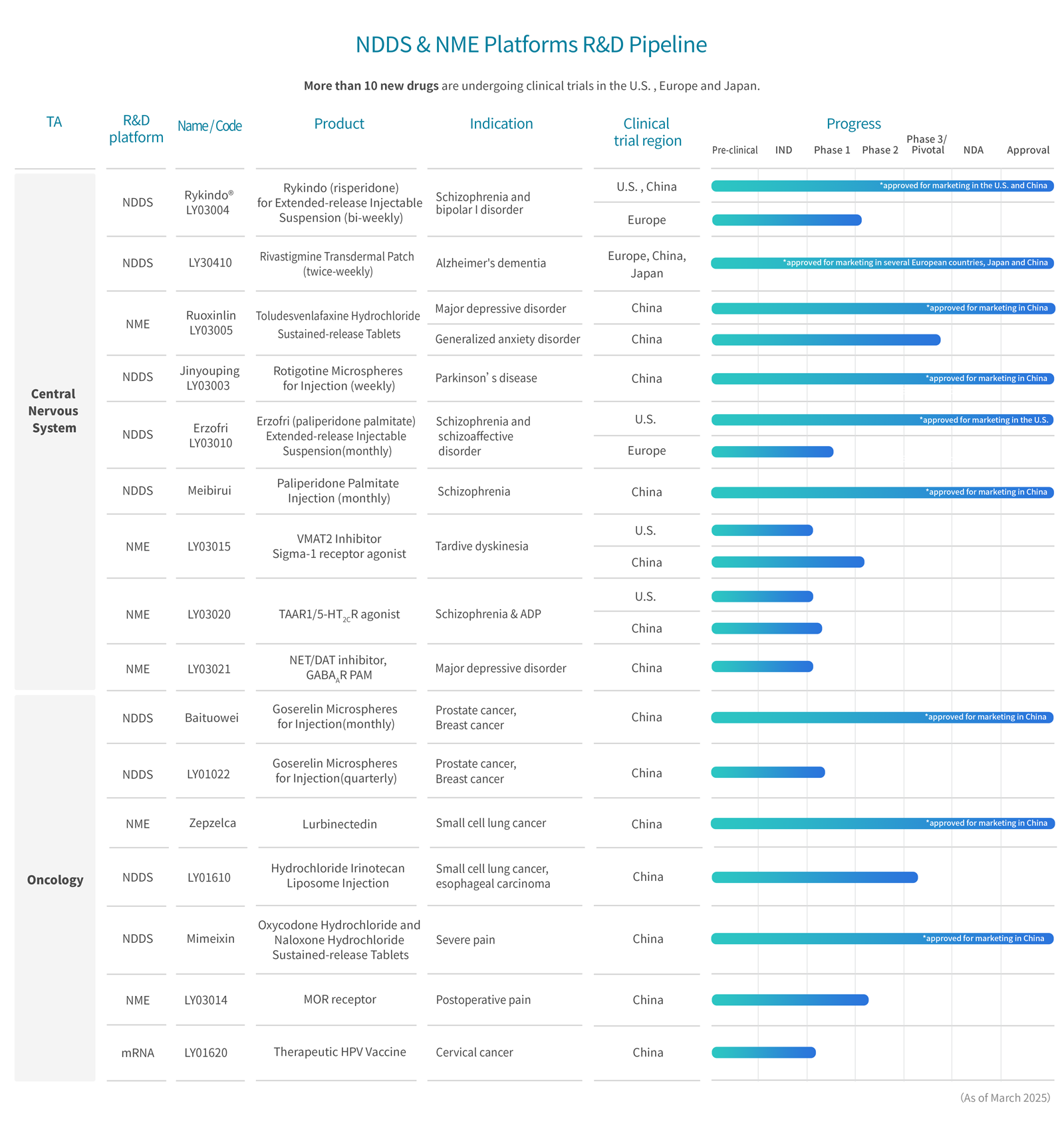
R&D Pipelines